THE KOJAK AUTO-DISABLE HYPODERMIC SYRINGE
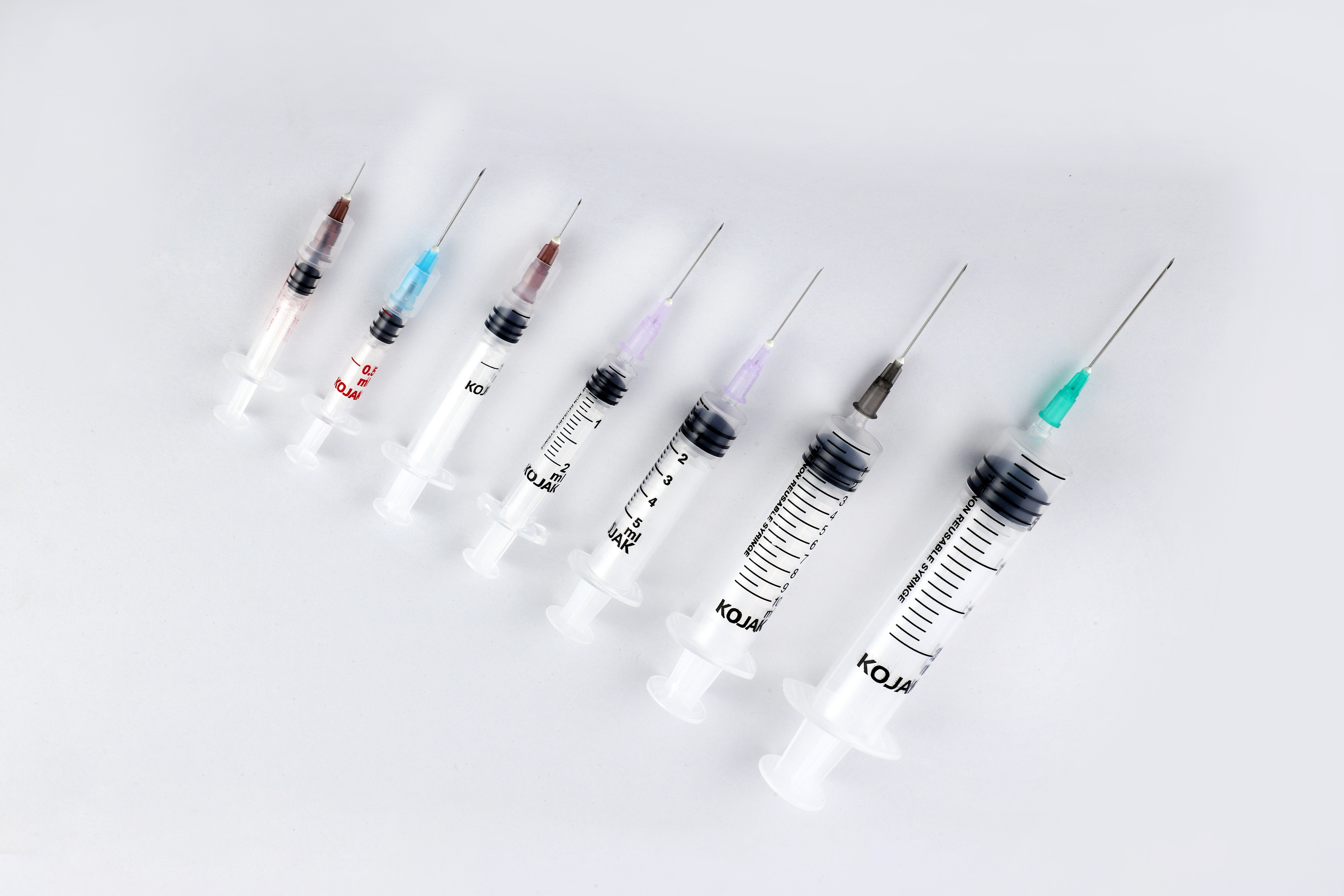
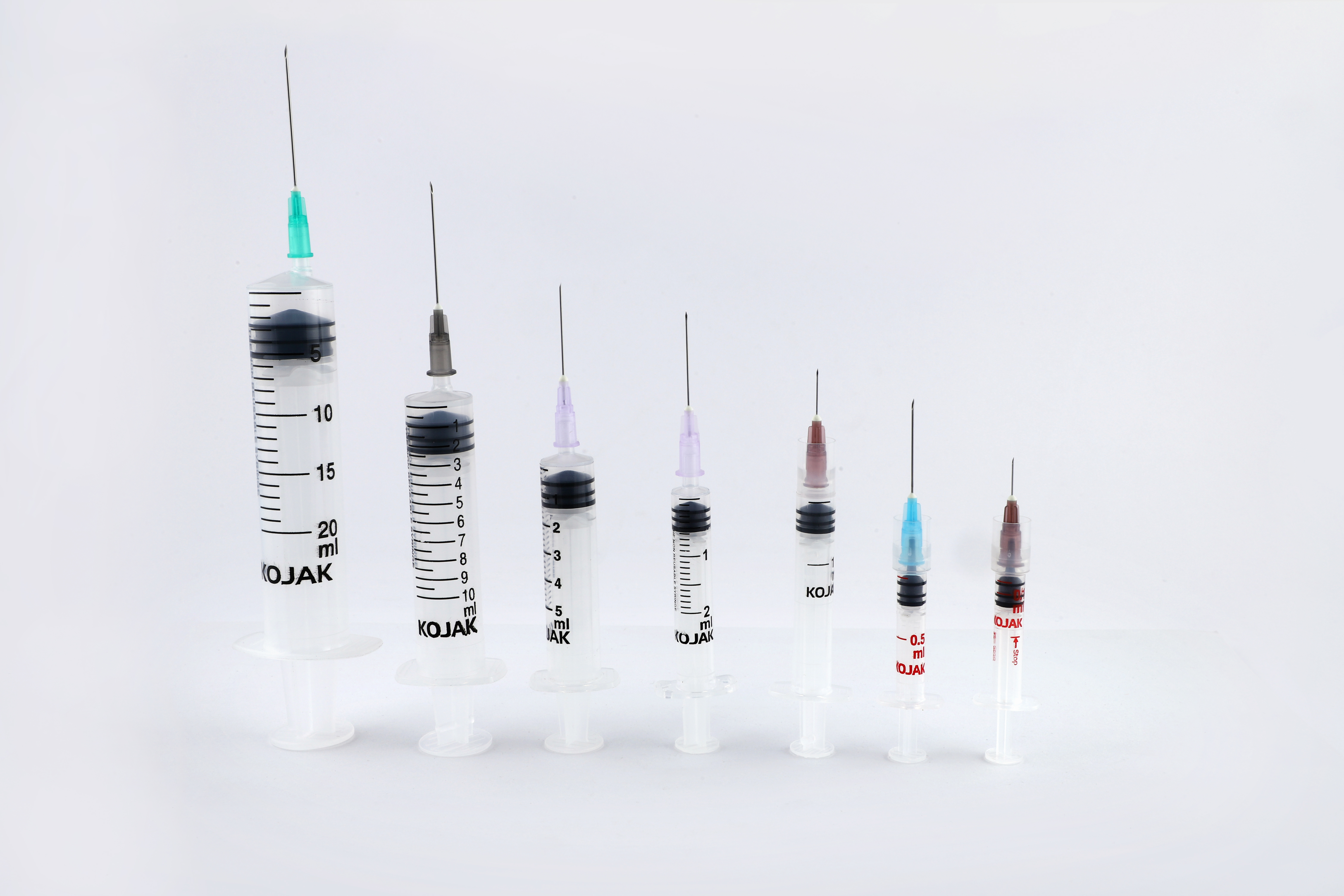
HMD Ltd. introduced the AD syringe in India in 2001, becoming the first auto disable syringe manufacturers in the country. It was launched in technical collaboration with United Kingdom-based global licensing company Star Syringes, which had patented the design. Sold under the brand name Kojak Selinge. It is available in the curative healthcare segment for various usage – in 2 ml, 3 ml, 5 ml, 10 ml, and 20 ml sizes. For immunization, it is available in 0.1 ml, 0.3ml, 0.5 ml, and 1 ml sizes. Kojak Selinge works on a ring & break mechanism which makes sure that the plunger breaks if re-use is attempted. An affordably priced syringe, it is available with any gauge of needle that is preferred by the user. Both detachable and fixed needle design options are present. Additional safety is ensured through the Luer-Lok tip – which enables the needle to be locked in place – and Luer slip tip – which enables a quicker fit. Auto-disable syringes are playing a major role in preventing and combating the spread of COVID-19, the deadly pandemic that has gripped the world since 2020. From the very beginning, WHO suggested the use of auto-disable syringes for collecting blood samples of Coronavirus patients, in order that the disease is not transmitted via healthcare equipment. Further, being recommended for giving vaccines in public immunization programmes, it is also playing a critical role in administrating the COVID-19 vaccine. The largest manufacturer of auto-disable syringes in the world, HMD Ltd. plans to scale up its production capacity from 700 million AD Syringes a year to one billion by mid-2021 as part of its commitment to produce an adequate number of AD syringes for the COVID-19 vaccination effort. One of the largest suppliers of auto-disable syringes to UNICEF for its immunization programmes for more than a decade, HMD has received orders for multiple syringes from UNICEF for the COVAX facility - a global initiative meant to accelerate the development and manufacture of COVID-19 vaccines, and to ensure that poorer nations have equitable access to it. At least 60 to 70 percent of the 1.3 billion people in India and 7.8 billion people in the world will need auto-disable syringes for the COVID-19 vaccine. Accordingly, we at HMD plan to allocate 50 percent of the total 0.3ML and 0.5 ML AD syringes for India and 50 percent for export to other countries, in keeping with our vision to contribute to affordable access and patient safety everywhere.